Energy And Place
How does energy production and consumption impact place?
How does your sense of place, environmental ethic and understanding of our energy needs influence your perception and decisions relating to energy production and consumption?
Scientific Paper
Conducting and Observing a Production of Biodiesel
Charlie Malone, Al Thomson, Aubree Lorenzen
Animas High School
Abstract: Biodiesel is a renewable, cleaner alternative to regular diesel. The importance behind making it was to understand how to produce biodiesel from vegetable oil and understand what type of reaction it undergoes to form the final substance. Originally, the experiment required the creation of crude biodiesel but after a few modifications, we changed it into filtered biodiesel. This reaction is also known as transesterification. Transesterification is the conversion of a carboxylic acid ester into a different carboxylic acid ester. Usually, these reactions involve alcohol. To create the biodiesel we had to use a fume-hood as the methanol is toxic and flammable, graduated cylinders to measure out the amount of a substance we needed. Then we combined the methanol and potassium hydroxide in a ball jar and allowed it to pressurize. Eventually, the substance was transferred to a separatory funnel where the glycerin and triglycerides were separated. Because of the process of the experiment, there were no official results, just observations recorded such as exothermic reactions and color changes. As far as problems on the process went, there were no known issues besides time management. However, even with time being short in the lab, we were successful because we were diligent about the jobs each of us was responsible for.
Introduction: The purpose of this lab was to create crude biodiesel from scratch using vegetable oil, methanol, and potassium hydroxide. This experiment was being done to understand how to produce biodiesel from scratch and what type of reaction it undergoes to further our knowledge of energy production. Also, we were curious about how crude biodiesel relates to normal diesel and the positive effects it has on the environment compared to the negative effects. The experiment undergoes a transesterification reaction, which is the conversion of a carboxylic acid ester into a different carboxylic acid ester. This means that a certain type of alcohol was combined with an acid to release an ester through an exothermic reaction. In this case, we mixed potassium hydroxide and methanol in a separatory funnel underneath a fume-hood.
Biodiesel is a vegetable oil or animal fat based diesel fuel. Biodiesel is the first EPA advanced
biofuel in commercial-scale production, created to be used in a standard diesel engine.
Its production has gained popularity because it is a renewable, clean burning diesel that reduces greenhouse gas emissions by 57-86% when compared to standard diesel. When used as a biofuel in vehicles biodiesel is usually in blends of 5-20% biodiesel.
A typical molecule of biodiesel is a long string of carbon and hydrogen atoms with an ester functional group at one end.
Biodiesel is chemically similar to regular diesel, however standard diesel lacks the ester group at the end shown above in blue.
To create biodiesel molecules, methanol (CH3OH) was mixed with vegetable oil. A molecule of vegetable oil is similar to biodiesel, however much larger. Notice, like biodiesel, it also has linked ester functional groups shown in blue.
Converting vegetable oil into biodiesel produces a transesterification reaction. In this reaction methanol (shown in green) is used to break the red bonds of the ester groups, making the molecules similar to regular diesel. Methoxy groups then replace the ester groups but are not linked vertically like a molecule of vegetable oil.
Methods:
1. Use a 50 ml-graduated cylinder to measure out 40 ml of methanol and pour it into a ball jar under a fume hood.
2. Use a balance to measure out 1.6g of potassium hydroxide.
3. Pour the potassium hydroxide into the ball jar with the methanol under the fume hood.
4. Seal the jar and shake it vigorously until all the potassium hydroxide is dissolved into the methanol.
5. Unseal the pressurized jar under the fume hood.
6. Using a 500 ml graduated cylinder measure out 200 ml of vegetable oil.
7. Pour the vegetable oil into the ball jar and seal the lid.
8. Shake the ball jar vigorously for 10 minutes.
9. Pour the crude biodiesel into the separatory funnel and let it sit for 30 minutes until the glycerin separates from the triglyceride and settles at the bottom.
10. Drain the glycerin into a waste jar.
11. Using a spray bottle, spray the surface of the biodiesel so all the remaining glycerin and excess methanol floating on the surface is dissolved into the water..
12. Drain the remaining glycerin into a waste jar.
13. Rinse the ball jar and drain the biodiesel into it. Seal the jar.
Hazards: It is critical to perform the experiment under a fume-hood (local ventilation device) so that the toxins of the methanol remain in one area, not to be exposed to human contact until later use. Also, because of the substances included in creating biodiesel it can be highly flammable as well as present reactive hazards. Such reactive hazards include concentrated bases and acids from the materials present. Health problems may result if the proper safety equipment is not used such as changes in gene structures, lung and cardiac reactions like changes to electrocardiograms (electric activity of the heart), heart rate variability, blood pressure, and lung function. Gloves, closed toed shoes and long pants must be worn when producing biodiesel. You should always be wearing safety goggles in the case that the liquid hits your eyes. If it does, immediately locate your eye-wash station and rinse thoroughly; if it gets on your skin locate the body-wash station and pull the lever. If ingested, consult your nearest hospital as soon as possible.
Observations: KOH with Methanol was a slow exothermic reaction that releases gas, which created pressure inside the ball jar
Vegetable oil with Methanol and KOH was a slow reaction that started milky and then proceeded to turn yellow.
When the crude oil was in the separatory funnel, the crude glycerin settled at the bottom and was easily drained
Distilled water was finely sprayed on the surface and sank to the bottom, pulling excess glycerin down with it.
Discussion: The purpose of making biodiesel was to observe the chemical reactions in each step to learn about the overall process of its production. We expected to end up with a product that matched that of the description in our methods. For the most part we were successful. In our final product there was still a small amount of glycerin left over at the bottom of the ball jar. This is because we did not spray the crude biodiesel enough to allow for the glycerin, basic salts and the unreacted methanol to dissolve into the water. To fix this we should have repeated steps 11 and 12 multiple times until it was visually clear that there was absolutely no amount of excess waste left. For future processes of making biodiesel we would carry out the entire production on a much longer time scale. Special care would be taken when measuring out each chemical to ensure precise amounts are being mixed for the given steps.
On the industrial scale the process of making biodiesel is continuous as opposed to in batches. For our small-scale production we measured out the certain chemicals to produce one small batch of biodiesel. A industrial process consists of a constant flow of chemicals that ensures maximum efficiency for heat and distribution of chemicals. A continuous production also allows for the greatest purity of biodiesel since there is a constant flow of fluids through a system there is no excess waste that settles anywhere.
Infographic
How does your sense of place, environmental ethic and understanding of our energy needs influence your perception and decisions relating to energy production and consumption?
Scientific Paper
Conducting and Observing a Production of Biodiesel
Charlie Malone, Al Thomson, Aubree Lorenzen
Animas High School
Abstract: Biodiesel is a renewable, cleaner alternative to regular diesel. The importance behind making it was to understand how to produce biodiesel from vegetable oil and understand what type of reaction it undergoes to form the final substance. Originally, the experiment required the creation of crude biodiesel but after a few modifications, we changed it into filtered biodiesel. This reaction is also known as transesterification. Transesterification is the conversion of a carboxylic acid ester into a different carboxylic acid ester. Usually, these reactions involve alcohol. To create the biodiesel we had to use a fume-hood as the methanol is toxic and flammable, graduated cylinders to measure out the amount of a substance we needed. Then we combined the methanol and potassium hydroxide in a ball jar and allowed it to pressurize. Eventually, the substance was transferred to a separatory funnel where the glycerin and triglycerides were separated. Because of the process of the experiment, there were no official results, just observations recorded such as exothermic reactions and color changes. As far as problems on the process went, there were no known issues besides time management. However, even with time being short in the lab, we were successful because we were diligent about the jobs each of us was responsible for.
Introduction: The purpose of this lab was to create crude biodiesel from scratch using vegetable oil, methanol, and potassium hydroxide. This experiment was being done to understand how to produce biodiesel from scratch and what type of reaction it undergoes to further our knowledge of energy production. Also, we were curious about how crude biodiesel relates to normal diesel and the positive effects it has on the environment compared to the negative effects. The experiment undergoes a transesterification reaction, which is the conversion of a carboxylic acid ester into a different carboxylic acid ester. This means that a certain type of alcohol was combined with an acid to release an ester through an exothermic reaction. In this case, we mixed potassium hydroxide and methanol in a separatory funnel underneath a fume-hood.
Biodiesel is a vegetable oil or animal fat based diesel fuel. Biodiesel is the first EPA advanced
biofuel in commercial-scale production, created to be used in a standard diesel engine.
Its production has gained popularity because it is a renewable, clean burning diesel that reduces greenhouse gas emissions by 57-86% when compared to standard diesel. When used as a biofuel in vehicles biodiesel is usually in blends of 5-20% biodiesel.
A typical molecule of biodiesel is a long string of carbon and hydrogen atoms with an ester functional group at one end.
Biodiesel is chemically similar to regular diesel, however standard diesel lacks the ester group at the end shown above in blue.
To create biodiesel molecules, methanol (CH3OH) was mixed with vegetable oil. A molecule of vegetable oil is similar to biodiesel, however much larger. Notice, like biodiesel, it also has linked ester functional groups shown in blue.
Converting vegetable oil into biodiesel produces a transesterification reaction. In this reaction methanol (shown in green) is used to break the red bonds of the ester groups, making the molecules similar to regular diesel. Methoxy groups then replace the ester groups but are not linked vertically like a molecule of vegetable oil.
Methods:
1. Use a 50 ml-graduated cylinder to measure out 40 ml of methanol and pour it into a ball jar under a fume hood.
2. Use a balance to measure out 1.6g of potassium hydroxide.
3. Pour the potassium hydroxide into the ball jar with the methanol under the fume hood.
4. Seal the jar and shake it vigorously until all the potassium hydroxide is dissolved into the methanol.
5. Unseal the pressurized jar under the fume hood.
6. Using a 500 ml graduated cylinder measure out 200 ml of vegetable oil.
7. Pour the vegetable oil into the ball jar and seal the lid.
8. Shake the ball jar vigorously for 10 minutes.
9. Pour the crude biodiesel into the separatory funnel and let it sit for 30 minutes until the glycerin separates from the triglyceride and settles at the bottom.
10. Drain the glycerin into a waste jar.
11. Using a spray bottle, spray the surface of the biodiesel so all the remaining glycerin and excess methanol floating on the surface is dissolved into the water..
12. Drain the remaining glycerin into a waste jar.
13. Rinse the ball jar and drain the biodiesel into it. Seal the jar.
Hazards: It is critical to perform the experiment under a fume-hood (local ventilation device) so that the toxins of the methanol remain in one area, not to be exposed to human contact until later use. Also, because of the substances included in creating biodiesel it can be highly flammable as well as present reactive hazards. Such reactive hazards include concentrated bases and acids from the materials present. Health problems may result if the proper safety equipment is not used such as changes in gene structures, lung and cardiac reactions like changes to electrocardiograms (electric activity of the heart), heart rate variability, blood pressure, and lung function. Gloves, closed toed shoes and long pants must be worn when producing biodiesel. You should always be wearing safety goggles in the case that the liquid hits your eyes. If it does, immediately locate your eye-wash station and rinse thoroughly; if it gets on your skin locate the body-wash station and pull the lever. If ingested, consult your nearest hospital as soon as possible.
Observations: KOH with Methanol was a slow exothermic reaction that releases gas, which created pressure inside the ball jar
Vegetable oil with Methanol and KOH was a slow reaction that started milky and then proceeded to turn yellow.
When the crude oil was in the separatory funnel, the crude glycerin settled at the bottom and was easily drained
Distilled water was finely sprayed on the surface and sank to the bottom, pulling excess glycerin down with it.
Discussion: The purpose of making biodiesel was to observe the chemical reactions in each step to learn about the overall process of its production. We expected to end up with a product that matched that of the description in our methods. For the most part we were successful. In our final product there was still a small amount of glycerin left over at the bottom of the ball jar. This is because we did not spray the crude biodiesel enough to allow for the glycerin, basic salts and the unreacted methanol to dissolve into the water. To fix this we should have repeated steps 11 and 12 multiple times until it was visually clear that there was absolutely no amount of excess waste left. For future processes of making biodiesel we would carry out the entire production on a much longer time scale. Special care would be taken when measuring out each chemical to ensure precise amounts are being mixed for the given steps.
On the industrial scale the process of making biodiesel is continuous as opposed to in batches. For our small-scale production we measured out the certain chemicals to produce one small batch of biodiesel. A industrial process consists of a constant flow of chemicals that ensures maximum efficiency for heat and distribution of chemicals. A continuous production also allows for the greatest purity of biodiesel since there is a constant flow of fluids through a system there is no excess waste that settles anywhere.
Infographic
Reflection
-What were you trying to convey to your audience through the creation of your infographic?
Explain your rationale for focusing on this particular topic/question.
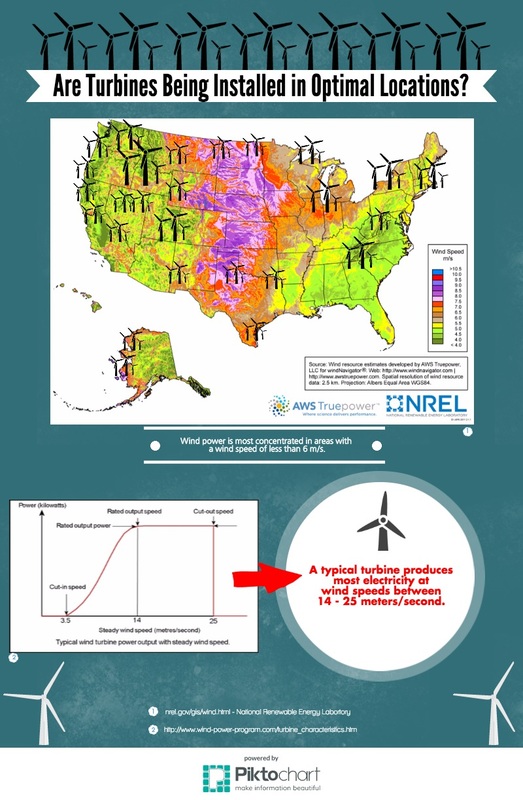
My infographic was about wind and how it is utilized as electricity here in the US. I correlated national average wind speeds with the concentration of electricity producing turbines. I also included a graph that explained the optimal wind speed for a turbine and as well as the maximum speed. I hoped to convey to my audience that areas with the highest concentration of wind turbines didn't have the most optimal windspeed. I also hoped to point out that the areas with the highest wind-speeds such as Texas and Arkansas have some of the fewest wind mills. This left the audience curious about the reason these areas had less turbines than places like California.
-What did you learn about energy or energy resources from your particular scientific investigation?
-What were you trying to convey to your audience through the creation of your infographic?
Explain your rationale for focusing on this particular topic/question.
My infographic was about wind and how it is utilized as electricity here in the US. I correlated national average wind speeds with the concentration of electricity producing turbines. I also included a graph that explained the optimal wind speed for a turbine and as well as the maximum speed. I hoped to convey to my audience that areas with the highest concentration of wind turbines didn't have the most optimal windspeed. I also hoped to point out that the areas with the highest wind-speeds such as Texas and Arkansas have some of the fewest wind mills. This left the audience curious about the reason these areas had less turbines than places like California.
-What did you learn about energy or energy resources from your particular scientific investigation?
Materials project
In this project we studied how chemistry has shaped our past, present, and may shape our future. I chose to research biodegradable plastics, specifically OXO biodegradable. It was interesting to see how the structure of matter on the molecular level directly determines a polymer's properties.
Plastic Engineered To Fail
Al Thomson
With global warming posing more of a threat each year, scientists have been put to the task of creating environmentally friendly materials. America has become dependent on the common polymer known as plastic. From computers to shopping bags, polymers are used everyday by most people.
Unfortunately for our planet, plastic takes hundreds of years to decompose. Plastic is being stockpiled in landfills and not going anywhere. According to research done by the University of Utah, enough plastic is thrown away each year to circle the earth four times.
These outrageous numbers will soon be dropping due to huge advancements that have been made in the plastic department. Scientists have essentially created a plastic that will decompose after 2-18 months. This plastic is called OXO-biodegradable plastic.
OXO-biodegradation is defined by CEN (the European Standards Organization) as “degradation resulting from oxidative and cell mediated phenomena, either simultaneously or successive.” OXO-biodegradable plastic is conventional plastic with small amounts of metal salt additive. The additive alters the natural degradation process to accelerate it. The OXO plastic will degrade at the end of its required lifetime in the presence of oxygen.
At the end of the process the material is no longer visible, it is also no longer a plastic. During the degradation process the plastic is converted from long chain polymers to small-chain chemicals, which will then biodegrade and turn into carbon dioxide, water, and biomass (cell structure of micro-organisms), leaving no traces of plastic in the environment.
The chemical degradation process for plastic is a reaction between the very long carbon and hydrogen polymer chains, and the oxygen in the air. For standard polymers like plastic the carbon and hydrogen molecules are very stable because the carbon atoms have four bonds while the hydrogen atoms have one. This results in a very stable material that takes hundreds of years to decompose because there is no place in the molecule for a reaction to take place.
The metal salt additive found in oxo-biodegradable plastic is called TDPA. When added to standard molecules in quantities ranging from 1%-5%, the additive is spaced throughout the long hydrogen and carbon chains breaking them up and making short enough molecules to react with oxygen and start to decompose.
The rate of decomposition is determined by the amount of additive in the polymer. This makes the plastics ‘programmable’. For example, a polymer with a concentration of one or two percent TDPA will biodegrade after several years while a polymer with five percent additive can biodegrade in just a couple weeks. When exposed to the elements TDPA incorporated polymers will fragment and biodegrade into carbon dioxide, water, and biomass.
This technology has been well known to polymer scientist for years. Manufactures of polymers usually add antioxidant chemicals into their materials to prevent oxidation. OXO-biodegradable polymers rely on the ability to manage these additives in a predictable way. Manufacturers aim to balance the effects of the TDPA additive with the effects of antioxidants to create products that satisfy needs long, useful shelf and service lives while creating biodegradation rates suitable for the disposal environment.
These polymers could replace many existing polymer products that currently are made from traditional methods. This would create shopping bags that would be used once or twice and disappear after the required job is done.
Elevator Pitch
Did you know that 50% of the plastic used today is thrown away after just one use? The best solution I have found is Oxo-biodegradable plastics. Oxo plastics are made with a small percent of additive that causes the plastics to break down and decompose after a desired time period. During the degradation process the plastic is converted from long chain polymers to small-chain chemicals, which will then biodegrade and turn into carbon dioxide, water, and biomass (cell structure of micro-organisms), leaving no traces of plastic in the environment. This could mean that the plastic that is only used once can be altered to completely disappear after a matter of weeks. I need funding to further my research on Oxo plastics. Oxo plastics have the potential to completely replace disposable packaging that is used just once. The process is easy and inexpensive however more research needs to be done. Factory tests should be done to ensure an uninterrupted shelf life. Experiments should also be conducted to evaluate and compare the properties of oxo plastics to common plastics.
Plastic Engineered To Fail
Al Thomson
With global warming posing more of a threat each year, scientists have been put to the task of creating environmentally friendly materials. America has become dependent on the common polymer known as plastic. From computers to shopping bags, polymers are used everyday by most people.
Unfortunately for our planet, plastic takes hundreds of years to decompose. Plastic is being stockpiled in landfills and not going anywhere. According to research done by the University of Utah, enough plastic is thrown away each year to circle the earth four times.
These outrageous numbers will soon be dropping due to huge advancements that have been made in the plastic department. Scientists have essentially created a plastic that will decompose after 2-18 months. This plastic is called OXO-biodegradable plastic.
OXO-biodegradation is defined by CEN (the European Standards Organization) as “degradation resulting from oxidative and cell mediated phenomena, either simultaneously or successive.” OXO-biodegradable plastic is conventional plastic with small amounts of metal salt additive. The additive alters the natural degradation process to accelerate it. The OXO plastic will degrade at the end of its required lifetime in the presence of oxygen.
At the end of the process the material is no longer visible, it is also no longer a plastic. During the degradation process the plastic is converted from long chain polymers to small-chain chemicals, which will then biodegrade and turn into carbon dioxide, water, and biomass (cell structure of micro-organisms), leaving no traces of plastic in the environment.
The chemical degradation process for plastic is a reaction between the very long carbon and hydrogen polymer chains, and the oxygen in the air. For standard polymers like plastic the carbon and hydrogen molecules are very stable because the carbon atoms have four bonds while the hydrogen atoms have one. This results in a very stable material that takes hundreds of years to decompose because there is no place in the molecule for a reaction to take place.
The metal salt additive found in oxo-biodegradable plastic is called TDPA. When added to standard molecules in quantities ranging from 1%-5%, the additive is spaced throughout the long hydrogen and carbon chains breaking them up and making short enough molecules to react with oxygen and start to decompose.
The rate of decomposition is determined by the amount of additive in the polymer. This makes the plastics ‘programmable’. For example, a polymer with a concentration of one or two percent TDPA will biodegrade after several years while a polymer with five percent additive can biodegrade in just a couple weeks. When exposed to the elements TDPA incorporated polymers will fragment and biodegrade into carbon dioxide, water, and biomass.
This technology has been well known to polymer scientist for years. Manufactures of polymers usually add antioxidant chemicals into their materials to prevent oxidation. OXO-biodegradable polymers rely on the ability to manage these additives in a predictable way. Manufacturers aim to balance the effects of the TDPA additive with the effects of antioxidants to create products that satisfy needs long, useful shelf and service lives while creating biodegradation rates suitable for the disposal environment.
These polymers could replace many existing polymer products that currently are made from traditional methods. This would create shopping bags that would be used once or twice and disappear after the required job is done.
Elevator Pitch
Did you know that 50% of the plastic used today is thrown away after just one use? The best solution I have found is Oxo-biodegradable plastics. Oxo plastics are made with a small percent of additive that causes the plastics to break down and decompose after a desired time period. During the degradation process the plastic is converted from long chain polymers to small-chain chemicals, which will then biodegrade and turn into carbon dioxide, water, and biomass (cell structure of micro-organisms), leaving no traces of plastic in the environment. This could mean that the plastic that is only used once can be altered to completely disappear after a matter of weeks. I need funding to further my research on Oxo plastics. Oxo plastics have the potential to completely replace disposable packaging that is used just once. The process is easy and inexpensive however more research needs to be done. Factory tests should be done to ensure an uninterrupted shelf life. Experiments should also be conducted to evaluate and compare the properties of oxo plastics to common plastics.